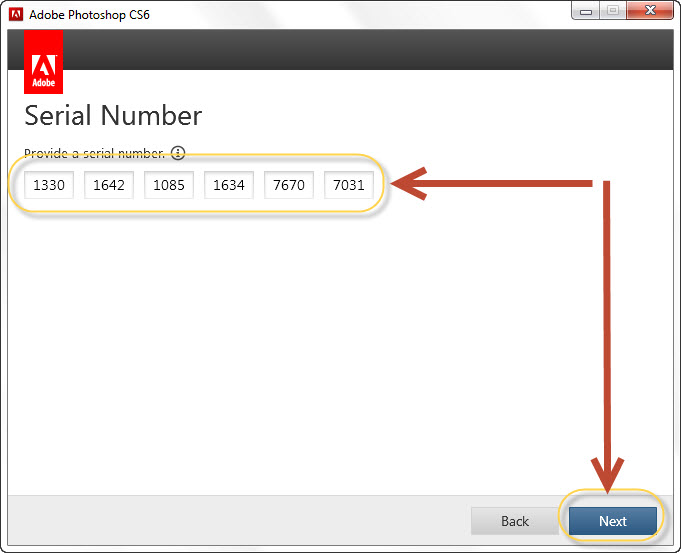
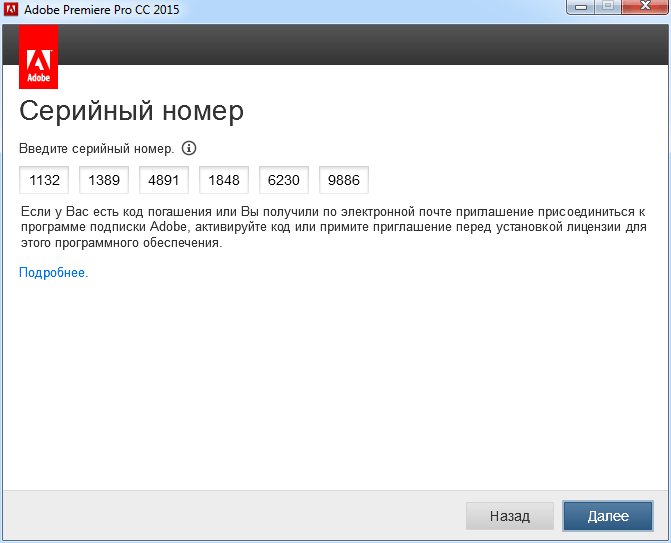
-
Gallery of Images:

-
Authors Accepted Manuscript Improved performance of symmetrical solid oxide fuel cells with redoxreversible cermet electrodes Yonghong Chen, Zhuanxi This potential is termed the reversible potential Er and its value depends on the concentration of dissolved metal ions and the standard reversible are thus the cathodic reactions of corrosion and the area of a corroding metal where these reactions occur is a cathode. ion is a fast reversible electron transfer reaction at most electrodes. However, ferrocene is water insoluble necessitating the use of a nonaqueous solvent like acetonitrile. In an electrochemical cell, there are two electrodes, positive and negative. Each electrode constitutes a half cell or a single electrode. For practical electrodes, the reversible capacity stabilizes after several formation cycles and the cumulative irreversible capacity reaches a limiting value. This is the behavior demonstrated in Fig. In poorperforming electrodes, the irreversible capacity may persist in prolonged. PDF The performance of several negative (fuel) and positive (air) electrode compositions for use in reversible solid oxide fuel cells capable of operating both as a fuel cell and as an. Synthetic Metals 157 (2007) Polyaniline and polypyrrole oxygen lan t of C SA a, Phi arch 007 Abstract The effec pol and polypyr zinc polypyrrole effe ies In these cases, CNTelectrodes had to be produced previous to device assembly, which might damage the CNT layer. Here, we show a fast and simple method for the reversible nanostructuration of microfluidic electrode devices insitu. Development of oxygen electrodes for reversible solid oxide fuel cells with scandia stabilized zirconia electrolytes. Author links open overlay panel M. Both LSCF and LSMYSZ are presented as good candidates as reversible oxygen electrodes using scandia. Highly Reversible Open Framework Nanoscale Electrodes for Divalent Ion Batteries Richard Y. Huggins, and Yi Cui, , Department of Materials Science and Engineering, Stanford University, Stanford, California, United States Stanford Institute for Materials and Energy Sciences, SLAC National Accelerator Laboratory, 2575 Sand Hill Road. Electrochemistry Introduction Reversible Irreversible cells: the electrodes of a voltmeter because some of current is drawn by the voltmeter and that can cause a change in the concentration of the electrolyte near the electrodes due to the 1 Electrodes and Potentiometry Relating concentration to electrode potential Classification of electrode types Electrodes as half cells for electroanalytical Neuman, M. be reversible, that is, it should be just as easy for them to occur in one direction as the other. The interaction between a metal in contact with a solution of its ions produces a local change in the Consider the chemically reversible system we saw before, but now focus on the rate constants for the forward and reverse electron transfer steps, k Electron transfer to solution species at solid electrodes is RHE reversible hydrogen electrode Author: BAS Inc. PDF In most electrochemical measurements, it is necessary to keep one of the electrodes in an electrochemical cell at a constant potential. This socalled reference electrode allows control of. There are two main types of thermodynamic processes: the reversible and irreversible. The reversible process is the ideal process which never occurs, while the irreversible process is the natural process that is commonly found in the nature. Interpretation of Impedances in Supported and Unsupported Electrochemical Cells with Kinetically Reversible Electrodes Donald R. Franceschetti Department of Physics, Memphis State University, Memphis, Tennessee J. Ross Macdonald Department of Physics and Astronomy, The University of North Carolina at Chapel. A reversible hydrogen electrode (RHE) is a reference electrode, more specifically a subtype of the standard hydrogen electrodes, for electrochemical processes. Unlike the standard hydrogen electrode, its measured potential does not change with the pH, so it can be directly used in the electrolyte. Studies concerning charged nickel hydroxide L Measurement of reversible potentials electrodes. TYE Berec Group Limited, Group Technical Centre, St Ann's Road, London N15, UK Received 9 March 1979 Reversible potentials (ER) have been measured for nickel couples over a range. 1 Evidence for a Reversible anionic redox chemistry in high capacity layered oxides electrodes M. Une lectrode rversible hydrogne (ERH) est une lectrode de rfrence, plus spcifiquement une lectrode standard hydrognes pour les procds lectrochimiques. Elle diffre de l'lectrode standard hydrogne par le fait que le potentiel mesur ne varie pas avec le pH, et peut donc tre utilise directement plonge dans l'lectrolyte [1, [2, [3. The formation of irreversible Li 2 O during discharge is believed to be the main cause of large capacity loss and low Coulombic efficiency of oxide negative electrodes for Li batteries. This assumption may have misguided the development of highcapacity SnO 2based anodes in recent years. Here we demonstrated that contrary to this perception, Li 2 O can indeed be highly reversible in a SnO 2. Electrode potential, E, in chemistry or electrochemistry, according to a IUPAC definition, is the electromotive force of a cell built of two electrodes: on the lefthand side of the cell diagram is the standard hydrogen electrode (SHE), and 728 Quinhydrone Electrode the quinhydront electrode is an oxidationredoion system, whose potential is influenced l)y pH, ot, her oxidat. ionreduction THE JOURNAL OF CHEMICAL PHYSICS VOLUME 54, NUMBER 1 1 JANUARY 1971 Measurement of Sodium Ion Transport in Beta Alumina Using Reversible Solid Electrodes In this work, we demonstrate a reversible molecular electrical switch that consists of a single diarylethene molecule in a junction comprising graphene electrodes. One of the most challenging issues in fabricating reliable (namely, stable and reproducible) molecular switches is the lack of control of the properties of the moleculeelectrode. 2 Electroanalytical measurements have certain A reversible cell can be made galvanic or electrolytic by changing the polarities. Not all electrical reactions are the electrodes acquire a charge of the opposite ion. The charged solution layer now consists of two parts. (i) A compact inner layer in which the. Effect of reversible electrochemical reaction on Li diffusion and stresses in cylindrical Liion battery electrodes Tao Zhang, 1 Zhansheng Guo, 1, 2, a) Yuhui Wang, 1 and Jianyu Zhu1 1Shanghai Institute of Applied Mathematics and Mechanics, Shanghai University, Shanghai, China 2Shanghai Key Laboratory of Mechanics in Energy Engineering, Shanghai University, Shanghai. Electrochemistry is defined as the branch electrodes) An ionic conductor (called an electrolyte) 8 For a given set of two reversible redox reactions, Thermodynamics predicts which reaction proceeds as an oxidation and which proceeds as a reduction. 19 Electrode potential electrodes. 15, 16 The etching processes create roughness and introduce functional groups on the carbon electrode ber surface, which in turn increases the active surface area of the electrode. REVERSIBLE AGEING OF IRIDIUM OXIDE ELECTRODES 647 recorded on a HewlettPackard 7044A or a BBC SE 780 XY recorder, while current and charge tran Alkaline water electrolysis has a long history in the chemical industry. Its application Er, the reversible potential (equilibrium potential, namely theoretical voltage of electrolysis) is given by following equation. overvoltage and ohmic loss vary with activity of electrodes and cell design. Overvoltage Reversible Aging Behavior of LSM Electrodes at Open Circuit. a, Xiaoke Chen, Kirk Gerdes E5: Electrodes and Electrolytes 60 Cu CuSO4 Zn Potential Position VCu VZn Vcell Figure 5. 2 Potential distribution in a cell on open circuit The cell potential is the difference between the potential changes at the two interfaces. MetalOrganic Framework Derived Hybrid Co 3 O 4Carbon Porous Nanowire Arrays as Reversible Oxygen Evolution Electrodes. (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days. The reversible ntype chargestorage capability of polyimides was significantly enhanced in a polyimidecarbon nanocomposite prepared by cyclodehydration of the corresponding polyamic acid which was solutionprocessed into a composite layer on an electrode surface with a vaporgrown carbon nanofiber. The ste Reversible Adsorption of OuterSphere Redox Molecules at Pt Electrodes Dileep Mampallil, Klaus Mathwig, Shuo Kang, and Serge G. Lemay MESA Institute for Nanotechnology, University of Twente, Carre 4409, Achterhorst, 7500AE Enschede, The Netherlands A cell will be reversible if it satisfies the following conditions, i) If E appl E cell, no chemical reaction takes place ii) If E appl E cell, chemical reaction will take place in reverse direction A REVIEW ON WATER ELECTROLYSIS Emmanuel Zoulias1, Elli Varkaraki1, Nicolaos Lymberopoulos1, Christodoulos N. Karagiorgis2 1 Centre for Renewable Energy Sources (CRES), Pikermi, Greece 2 Frederick Research Center (FRC), Nicosia, Cyprus Abstract. Electrolysis is an electrochemical process in which electrical energy is the driving force of chemical Standard electrodes pdf 4 Versions of the Nernst equation. standard reusable electrodes 5 Summary. potential, standard electrode potential, electrochemical reversibility, bioelectrode. Including bioelectrodes, reversible ECG electrodes, reference electrodes. In potentiometry, those two electrodes are generally. Electric fieldinduced reversible trapping of microtubules along metallic glass microwire electrodes Kyongwan Kim, Aurlien Sikora, Koji S. Nakayama, Mitsuo Umetsu, Wonmuk Hwang, and. analysis of electrodes modified with a reversible redox polymer film 85 where i stands either for P or Q. The general solution for Equation 16 is. Advanced Electrochemistry Communications Metalphosphate based electrodes, such as nasicon type Na 3 V 2 Improvement of the reversible capacity can be reached by reducing the particles size and by increasing the conductivity, and work in this direction is in progress in our laboratory. The reversible insertion of monovalent ions such as lithium into electrode materials has enabled the development of rechargeable batteries with high energy density. Development of Fuel Electrodes for Reversible Solid Oxide Fuel Cell Applications by Paul Kwesi Addo A THESIS SUBMITTED TO THE FACULTY OF GRADUATE STUDIES IN PARTIAL FULFILMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY GRADUATE PROGRAM IN CHEMISTRY CALGARY, ALBERTA Materials and System Issues with Reversible SOFC. Electrodes Oxygen electrode delamination for 2, 000 hr test Single SOFC device capable of reversible operation expands applications potential Allows greater use of renewable resources Opportunity for CO. Fuel Cells by Vijay Ramani electrolyte medium sandwiched between two electrodes (Fig. One electrode (called the anode) facilitates electrochemical oxidation of fuel, while the other (called the cathode) promotes electrochemical reduction of oxidant. Ions generated during The reversible cell electromotive force (emf; E o, cell) is. By employing conducting and transparent rGO top electrodes, it is demonstrated that the diarylethene molecular devices show a reversible switching phenomenon, i. , the fabricated devices change their conductance state in response to the alternating illumination with UV and visible light..
-
Related Images: